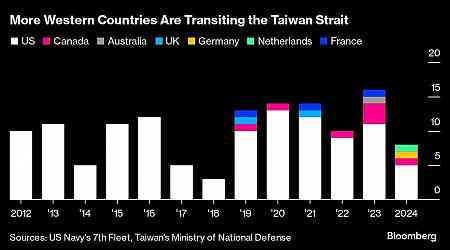
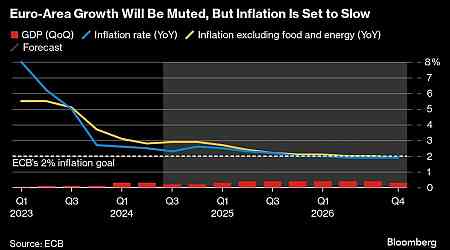
Financial Personal
Zambon Announces Publication of Phase 3 PROMIS I and II Study Results in The Lancet Respiratory Medicine Showing Efficacy Results of CMS I-neb in Patients with Non-Cystic Fibrosis Bronchiectasis Colonized with P. aeruginosa
Inhalation of colistimethate sodium via the I-neb® Adaptive Aerosol Delivery System (CMS I-neb) significantly reduced pulmonary exacerbations and improved quality of life in non-cystic fibrosis bronchiectasis (NCFB) patients colonized with P. aerugin
MILAN -- Zambon, a multinational pharmaceutical company focused on innovating cure and care to improve people’s health and the quality of patients’ lives, today announced the online publication of its Phase 3 PROMIS-I and PROMIS-II studies in The Lancet Respiratory Medicine journal. The PROMIS studies assessed the efficacy and safety of inhaled colistimethate sodium administered via the I-neb® Adaptive Aerosol Delivery System (“CMS I-neb”) as a treatment to reduce the frequency of pulmonary exacerbations in patients with Non-Cystic Fibrosis Bronchiectasis (NCFB) chronically colonized with P. aeruginosa over 12 months, compared to placebo. Read More